🤚🏻Factors Affecting Enzyme Activity and Enzyme Inhibitors
Effects of substrate concentration, temperature, pH, and hydration on enzyme activity, competitive vs non-competitive inhibition, feedback inhibition, and practical applications
From Field to Lab — When Enzymes Stop Working
In the previous lesson, we explored enzyme structure — what enzymes are, how the lock-and-key and induced-fit models explain specificity, and how cofactors and coenzymes assist catalysis. Now we turn to the practical question: what controls how fast an enzyme works?
A farmer stores wheat grain at 12% moisture and it remains viable for years — the enzymes inside are inactive because there is not enough water. Add water during sowing, and enzymes spring to life, breaking down starch into sugars that fuel germination. Now consider the opposite problem: a seed lot stored in a hot, humid warehouse. The high temperature and moisture activate enzymes prematurely, the seed “respires away” its food reserves, and germination rates plummet.
These real-world examples demonstrate that enzyme activity depends critically on substrate concentration, temperature, pH, hydration, and the presence of inhibitors. Understanding these factors is essential for both agriculture and biochemistry exams.
This lesson covers:
- Factors affecting enzyme activity — substrate concentration, enzyme concentration, temperature (Q₁₀), pH, hydration, and end-product effects
- Enzyme inhibitors — competitive vs non-competitive (reversible and irreversible), feedback inhibition
- Agricultural and medical applications — seed storage, sulpha drugs, cyanide poisoning
All sections are high-yield for IBPS AFO and ICAR exams.
Factors Affecting Enzyme Activity
Enzyme activity is not constant — it varies with the physical and chemical environment. Six major factors determine how fast an enzyme catalyses its reaction. Each has direct agricultural relevance, from seed storage to soil enzyme activity.
1. Substrate Concentration
- Increasing substrate concentration initially increases reaction rate
- When all enzyme active sites are occupied, the rate reaches a plateau called Vmax (maximum velocity)
- Beyond Vmax, adding more substrate has no effect — the enzyme is saturated

TIP
Agricultural parallel: Increasing fertiliser (substrate) initially boosts crop growth (enzyme activity), but beyond a point, adding more fertiliser gives no benefit — the plant’s metabolic machinery is “saturated.”
2. Enzyme Concentration
- Increasing enzyme concentration increases reaction rate — until substrate becomes the limiting factor
- This mirrors the relationship with substrate concentration: whichever is in limiting quantity controls the rate
3. Temperature
| Temperature | Effect on Enzymes |
|---|---|
| 0°C | Reaction rate = zero (enzymes inactive but not destroyed) |
| 10–30°C | Rate doubles for every 10°C rise (Q₁₀ = 2) |
| 30°C | Optimum temperature for most enzymes |
| 55–60°C | Denaturation — permanent loss of enzyme structure and function |
- Q₁₀ (temperature coefficient) = rate at (T+10°C) / rate at T°C = 2 for most enzymes
- Denaturation occurs because high temperatures disrupt hydrogen bonds and weak interactions that maintain the enzyme’s 3D structure
IMPORTANT
Exam fact: Optimum enzyme temperature is 30°C. At 0°C, enzymes are inactive but recoverable. Above 55–60°C, enzymes are permanently denatured.
4. pH (Hydrogen Ion Concentration)
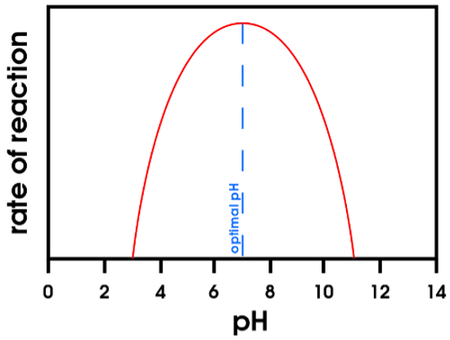
- Ideal pH for most enzymes: 7–7.5
- Exceptions exist: Pepsin works best at pH 1.5–3.0 (stomach acid); Trypsin works at high pH (alkaline intestine)
- pH changes alter the ionisation state of amino acids at the active site, affecting substrate binding
5. Hydration (Water Content)
- In dry seeds, water is too low for enzymatic activity → seeds remain dormant
- Adding water → enzymes become active → seed germinates
- Water serves as both solvent for enzyme-substrate interactions and a direct reactant in hydrolysis reactions
TIP
Agricultural application: Seed storage requires low moisture (8–12%) to keep enzymes inactive and prevent premature germination. This is why seeds must be dried before storage.
6. End Product Concentration
- Enzyme reactions are reversible (mass action principle)
- Accumulation of end products increases the rate of the reverse reaction
- In living cells, end products are continuously removed by subsequent metabolic reactions, keeping the forward reaction going
Enzyme Inhibitors
While the previous section covered physical factors (temperature, pH, water), enzymes can also be slowed or stopped by specific molecules that interfere with catalysis. These inhibitors are central to drug design (sulpha drugs, antibiotics), pesticide development, and the cell’s own metabolic self-regulation.
Enzyme inhibitors are molecules that reduce or prevent enzyme activity. The key distinction is where the inhibitor binds and whether the inhibition can be reversed.
Comparison of Inhibitor Types
| Feature | Competitive | Non-competitive (Irreversible) | Non-competitive (Reversible) |
|---|---|---|---|
| Binding site | Active site | Other site (destroys enzyme) | Allosteric site |
| Similarity to substrate | Structurally similar | Not similar | Not similar |
| Reversibility | Reversible (overcome by more substrate) | Irreversible (permanent damage) | Reversible (inhibitor can detach) |
| Effect on Vmax | Unchanged (if enough substrate) | Reduced | Reduced |
| Example | Malonate vs Succinate | CN⁻ (cyanide), CO, toxic metals | Feedback inhibition |
Competitive Inhibition
- Inhibitor is structurally similar to the substrate (substrate analogue)
- Competes for the same active site
- Can be overcome by increasing substrate concentration
- Example: Malonate inhibits Succinic dehydrogenase (malonate mimics succinate)
TIP
Medical application: Sulpha drugs are competitive inhibitors — they mimic PABA (p-aminobenzoic acid) and block bacterial folic acid synthesis, killing bacteria. Humans are unaffected because they obtain folic acid from diet.
Non-competitive Inhibition (Irreversible)
- Inhibitor binds at a different site from the active site
- Destroys the sulfhydryl (S-H) group of the enzyme
- Cannot be reversed by adding more substrate
- Examples: CN⁻ (cyanide), CO, toxic metals
WARNING
Cyanide poisoning works by irreversibly binding to cytochrome oxidase in the electron transport chain, blocking cellular respiration and ATP production entirely. This is why cyanide is lethal.
Non-competitive Inhibition (Reversible) — Feedback Inhibition
- Inhibitor binds at the allosteric site reversibly
- The end product of a metabolic pathway inhibits an earlier enzyme in the pathway
- Also called retro inhibition or feedback inhibition
- Example: Glucose-6-phosphate inhibits hexokinase
IMPORTANT
Feedback inhibition is one of the most important regulatory mechanisms in metabolism. The end product signals “enough has been made” and shuts down production — preventing wasteful overproduction.
Comparison Table — Competitive vs Non-competitive Inhibition
| Feature | Competitive | Non-competitive |
|---|---|---|
| Binds to | Active site | Other site or allosteric site |
| Structurally similar to substrate? | Yes | No |
| Overcome by excess substrate? | Yes | No |
| Examples | Malonate, Sulpha drugs | CN⁻, CO, feedback inhibitors |
| Km value | Increased (apparent) | Unchanged |
| Vmax | Unchanged | Decreased |
Summary Table — Key Facts at a Glance
| Fact | Answer |
|---|---|
| Q₁₀ value for enzymes | 2 (rate doubles per 10°C rise) |
| Optimum temperature | 30°C |
| Denaturation temperature | 55–60°C |
| Ideal pH for most enzymes | 7–7.5 |
| Pepsin optimal pH | 1.5–3.0 (highly acidic) |
| Seed dormancy due to | Low water → enzymes inactive |
| Competitive inhibitor example | Malonate (inhibits Succinic dehydrogenase) |
| Sulpha drugs inhibit | Bacterial folic acid synthesis (competitive) |
| Cyanide inhibits | Cytochrome oxidase (irreversible non-competitive) |
| Feedback inhibition = | End product inhibits earlier enzyme |
| Glucose-6-P inhibits | Hexokinase (feedback) |
| Reversible competitive overcome by | Increasing substrate concentration |
Summary Cheat Sheet
| Fact | Answer |
|---|---|
| Q₁₀ value for most enzymes | 2 (rate doubles per 10°C rise) |
| Optimum temperature for most enzymes | 30°C |
| Enzyme denaturation temperature | 55–60°C |
| At 0°C, enzymes are | Inactive but not destroyed (recoverable) |
| Ideal pH for most enzymes | 7–7.5 |
| Pepsin optimal pH | 1.5–3.0 (stomach acid) |
| Trypsin optimal pH | Alkaline (pH ~8) (intestinal) |
| Vmax is reached when | All enzyme active sites are saturated with substrate |
| Seed storage moisture for dormancy | 8–12% (keeps enzymes inactive) |
| Classic competitive inhibitor example | Malonate inhibits Succinic dehydrogenase |
| Competitive inhibitor resembles | The substrate (structurally similar) |
| How to overcome competitive inhibition | Increase substrate concentration |
| Sulpha drugs mechanism | Competitive inhibitor mimicking PABA, blocks bacterial folic acid synthesis |
| Cyanide (CN⁻) inhibits | Cytochrome oxidase (irreversible non-competitive) |
| Irreversible inhibitors destroy | The sulfhydryl (S-H) group of the enzyme |
| Feedback inhibition is also called | Retro inhibition |
| Feedback inhibition example | Glucose-6-phosphate inhibits Hexokinase |
| Competitive inhibition: effect on Vmax | Unchanged (if enough substrate added) |
| Non-competitive inhibition: effect on Vmax | Decreased |
| pH changes affect enzymes by altering | Ionisation state of amino acids at the active site |
TIP
Next: Lesson 05-01 moves from biochemistry to the whole-plant level — Plant Growth, where enzyme-driven processes manifest as cell division, elongation, and differentiation.
Pro Content Locked
Upgrade to Pro to access this lesson and all other premium content.
₹2388 billed yearly
- All Agriculture & Banking Courses
- AI Lesson Questions (100/day)
- AI Doubt Solver (50/day)
- Glows & Grows Feedback (30/day)
- AI Section Quiz (20/day)
- 22-Language Translation (30/day)
- Recall Questions (20/day)
- AI Quiz (15/day)
- AI Quiz Paper Analysis
- AI Step-by-Step Explanations
- Spaced Repetition Recall (FSRS)
- AI Tutor
- Immersive Text Questions
- Audio Lessons — Hindi & English
- Mock Tests & Previous Year Papers
- Summary & Mind Maps
- XP, Levels, Leaderboard & Badges
- Generate New Classrooms
- Voice AI Teacher (AgriDots Live)
- AI Revision Assistant
- Knowledge Gap Analysis
- Interactive Revision (LangGraph)
🔒 Secure via Razorpay · Cancel anytime · No hidden fees
From Field to Lab — When Enzymes Stop Working
In the previous lesson, we explored enzyme structure — what enzymes are, how the lock-and-key and induced-fit models explain specificity, and how cofactors and coenzymes assist catalysis. Now we turn to the practical question: what controls how fast an enzyme works?
A farmer stores wheat grain at 12% moisture and it remains viable for years — the enzymes inside are inactive because there is not enough water. Add water during sowing, and enzymes spring to life, breaking down starch into sugars that fuel germination. Now consider the opposite problem: a seed lot stored in a hot, humid warehouse. The high temperature and moisture activate enzymes prematurely, the seed “respires away” its food reserves, and germination rates plummet.
These real-world examples demonstrate that enzyme activity depends critically on substrate concentration, temperature, pH, hydration, and the presence of inhibitors. Understanding these factors is essential for both agriculture and biochemistry exams.
This lesson covers:
- Factors affecting enzyme activity — substrate concentration, enzyme concentration, temperature (Q₁₀), pH, hydration, and end-product effects
- Enzyme inhibitors — competitive vs non-competitive (reversible and irreversible), feedback inhibition
- Agricultural and medical applications — seed storage, sulpha drugs, cyanide poisoning
All sections are high-yield for IBPS AFO and ICAR exams.
Factors Affecting Enzyme Activity
Enzyme activity is not constant — it varies with the physical and chemical environment. Six major factors determine how fast an enzyme catalyses its reaction. Each has direct agricultural relevance, from seed storage to soil enzyme activity.
1. Substrate Concentration
- Increasing substrate concentration initially increases reaction rate
- When all enzyme active sites are occupied, the rate reaches a plateau called Vmax (maximum velocity)
- Beyond Vmax, adding more substrate has no effect — the enzyme is saturated

TIP
Agricultural parallel: Increasing fertiliser (substrate) initially boosts crop growth (enzyme activity), but beyond a point, adding more fertiliser gives no benefit — the plant’s metabolic machinery is “saturated.”
2. Enzyme Concentration
- Increasing enzyme concentration increases reaction rate — until substrate becomes the limiting factor
- This mirrors the relationship with substrate concentration: whichever is in limiting quantity controls the rate
3. Temperature
| Temperature | Effect on Enzymes |
|---|---|
| 0°C | Reaction rate = zero (enzymes inactive but not destroyed) |
| 10–30°C | Rate doubles for every 10°C rise (Q₁₀ = 2) |
| 30°C | Optimum temperature for most enzymes |
| 55–60°C | Denaturation — permanent loss of enzyme structure and function |
- Q₁₀ (temperature coefficient) = rate at (T+10°C) / rate at T°C = 2 for most enzymes
- Denaturation occurs because high temperatures disrupt hydrogen bonds and weak interactions that maintain the enzyme’s 3D structure
IMPORTANT
Exam fact: Optimum enzyme temperature is 30°C. At 0°C, enzymes are inactive but recoverable. Above 55–60°C, enzymes are permanently denatured.
4. pH (Hydrogen Ion Concentration)

- Ideal pH for most enzymes: 7–7.5
- Exceptions exist: Pepsin works best at pH 1.5–3.0 (stomach acid); Trypsin works at high pH (alkaline intestine)
- pH changes alter the ionisation state of amino acids at the active site, affecting substrate binding
5. Hydration (Water Content)
- In dry seeds, water is too low for enzymatic activity → seeds remain dormant
- Adding water → enzymes become active → seed germinates
- Water serves as both solvent for enzyme-substrate interactions and a direct reactant in hydrolysis reactions
TIP
Agricultural application: Seed storage requires low moisture (8–12%) to keep enzymes inactive and prevent premature germination. This is why seeds must be dried before storage.
6. End Product Concentration
- Enzyme reactions are reversible (mass action principle)
- Accumulation of end products increases the rate of the reverse reaction
- In living cells, end products are continuously removed by subsequent metabolic reactions, keeping the forward reaction going
Enzyme Inhibitors
While the previous section covered physical factors (temperature, pH, water), enzymes can also be slowed or stopped by specific molecules that interfere with catalysis. These inhibitors are central to drug design (sulpha drugs, antibiotics), pesticide development, and the cell’s own metabolic self-regulation.
Enzyme inhibitors are molecules that reduce or prevent enzyme activity. The key distinction is where the inhibitor binds and whether the inhibition can be reversed.
Comparison of Inhibitor Types
| Feature | Competitive | Non-competitive (Irreversible) | Non-competitive (Reversible) |
|---|---|---|---|
| Binding site | Active site | Other site (destroys enzyme) | Allosteric site |
| Similarity to substrate | Structurally similar | Not similar | Not similar |
| Reversibility | Reversible (overcome by more substrate) | Irreversible (permanent damage) | Reversible (inhibitor can detach) |
| Effect on Vmax | Unchanged (if enough substrate) | Reduced | Reduced |
| Example | Malonate vs Succinate | CN⁻ (cyanide), CO, toxic metals | Feedback inhibition |
Competitive Inhibition
- Inhibitor is structurally similar to the substrate (substrate analogue)
- Competes for the same active site
- Can be overcome by increasing substrate concentration
- Example: Malonate inhibits Succinic dehydrogenase (malonate mimics succinate)
TIP
Medical application: Sulpha drugs are competitive inhibitors — they mimic PABA (p-aminobenzoic acid) and block bacterial folic acid synthesis, killing bacteria. Humans are unaffected because they obtain folic acid from diet.
Non-competitive Inhibition (Irreversible)
- Inhibitor binds at a different site from the active site
- Destroys the sulfhydryl (S-H) group of the enzyme
- Cannot be reversed by adding more substrate
- Examples: CN⁻ (cyanide), CO, toxic metals
WARNING
Cyanide poisoning works by irreversibly binding to cytochrome oxidase in the electron transport chain, blocking cellular respiration and ATP production entirely. This is why cyanide is lethal.
Non-competitive Inhibition (Reversible) — Feedback Inhibition
- Inhibitor binds at the allosteric site reversibly
- The end product of a metabolic pathway inhibits an earlier enzyme in the pathway
- Also called retro inhibition or feedback inhibition
- Example: Glucose-6-phosphate inhibits hexokinase
IMPORTANT
Feedback inhibition is one of the most important regulatory mechanisms in metabolism. The end product signals “enough has been made” and shuts down production — preventing wasteful overproduction.
Comparison Table — Competitive vs Non-competitive Inhibition
| Feature | Competitive | Non-competitive |
|---|---|---|
| Binds to | Active site | Other site or allosteric site |
| Structurally similar to substrate? | Yes | No |
| Overcome by excess substrate? | Yes | No |
| Examples | Malonate, Sulpha drugs | CN⁻, CO, feedback inhibitors |
| Km value | Increased (apparent) | Unchanged |
| Vmax | Unchanged | Decreased |
Summary Table — Key Facts at a Glance
| Fact | Answer |
|---|---|
| Q₁₀ value for enzymes | 2 (rate doubles per 10°C rise) |
| Optimum temperature | 30°C |
| Denaturation temperature | 55–60°C |
| Ideal pH for most enzymes | 7–7.5 |
| Pepsin optimal pH | 1.5–3.0 (highly acidic) |
| Seed dormancy due to | Low water → enzymes inactive |
| Competitive inhibitor example | Malonate (inhibits Succinic dehydrogenase) |
| Sulpha drugs inhibit | Bacterial folic acid synthesis (competitive) |
| Cyanide inhibits | Cytochrome oxidase (irreversible non-competitive) |
| Feedback inhibition = | End product inhibits earlier enzyme |
| Glucose-6-P inhibits | Hexokinase (feedback) |
| Reversible competitive overcome by | Increasing substrate concentration |
Summary Cheat Sheet
| Fact | Answer |
|---|---|
| Q₁₀ value for most enzymes | 2 (rate doubles per 10°C rise) |
| Optimum temperature for most enzymes | 30°C |
| Enzyme denaturation temperature | 55–60°C |
| At 0°C, enzymes are | Inactive but not destroyed (recoverable) |
| Ideal pH for most enzymes | 7–7.5 |
| Pepsin optimal pH | 1.5–3.0 (stomach acid) |
| Trypsin optimal pH | Alkaline (pH ~8) (intestinal) |
| Vmax is reached when | All enzyme active sites are saturated with substrate |
| Seed storage moisture for dormancy | 8–12% (keeps enzymes inactive) |
| Classic competitive inhibitor example | Malonate inhibits Succinic dehydrogenase |
| Competitive inhibitor resembles | The substrate (structurally similar) |
| How to overcome competitive inhibition | Increase substrate concentration |
| Sulpha drugs mechanism | Competitive inhibitor mimicking PABA, blocks bacterial folic acid synthesis |
| Cyanide (CN⁻) inhibits | Cytochrome oxidase (irreversible non-competitive) |
| Irreversible inhibitors destroy | The sulfhydryl (S-H) group of the enzyme |
| Feedback inhibition is also called | Retro inhibition |
| Feedback inhibition example | Glucose-6-phosphate inhibits Hexokinase |
| Competitive inhibition: effect on Vmax | Unchanged (if enough substrate added) |
| Non-competitive inhibition: effect on Vmax | Decreased |
| pH changes affect enzymes by altering | Ionisation state of amino acids at the active site |
TIP
Next: Lesson 05-01 moves from biochemistry to the whole-plant level — Plant Growth, where enzyme-driven processes manifest as cell division, elongation, and differentiation.
Knowledge Check
Take a dynamically generated quiz based on the material you just read to test your understanding and get personalized feedback.
Lesson Doubts
Ask questions, get expert answers